Neutral, polar and electrically charged particles
On the smallest scale, the brain, like all other material objects, is made out of atoms, which in turn are made out of positively charged atomic nuclei which are surrounded by a shell containing positively charged electrons.
On earth, and in our brains, atoms rarely float about in isolation, but often cluster together to form molecules, in which several atoms share some of their electrons to form a common, shared outer shell. Chemists call this linking of atoms through a shared electron shell a covalent bond.
Normally, the number of positive charges in the nuclei of an atom or molecule is equal to the number of electrons in its shell, and the molecule is then said to be electically neutral. The electrostatic forces emanating from the positively charged nucleus are exactly cancelled by the opposite forces from the negatively charged electrons.
However, there are two ways in which atoms or molecules can deviate from such a state of neutral balance of electic forces: they can either be polar, or they can become ionized.
In polar molecules, the electrons are not evenly distributed around the shell of the molecule. A very common example of such a molecule, and indeed the most abundant molecule in our brains, is water. Water (H2O) is made of one oxygen atom and two hydrogen atoms linked together through their shared outer electron shell.
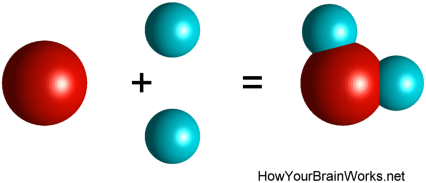
But the electrons that form the outer shell are more closely associated with the oxygen nucleus than with the hydrogen nuclei. The electric charge distribution along the water molecule is therefore not even, and the oxygen atom is slightly more negatively charged than the two hydrogen atoms. This contributes to the propensity of water molecules to hang together and form drops: the positive H end of one water molecule is electrically attracted to the O end of the next molecule.
Sometimes atoms acquire extra electrons in their shell, and thereby become electrically ... Ionized particles ... under construction
There are two fundamental driving forces underlying all electrical activity in the brain: electrostatic forces between

